Cell Culture Media Market Growth Factors
Cell Culture Media Market Growth & Trends
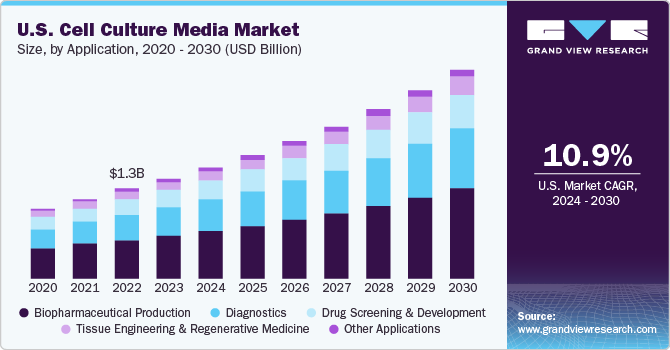
The global cell culture media market size is
expected to reach USD 10.2 billion by 2030, according to a new report by Grand
View Research, Inc. The market is expected to expand at a CAGR of 12.1% from
2022 to 2030. Expansion of biosimilars and biologics, growth in stem cell
research, and emerging bio manufacturing technologies for cell-based vaccines
are the major factors which are likely to drive the market. For instance, in
October 2021, the Australian Government funded the Australian-led stem cell
research through USD 25 million in grants.
The expansion of clear, regulatory approval paths for
biosimilars in emerging markets is generating great opportunities for
biosimilar monoclonal antibodies. The
availability of an approval pathway in the U.S., has led to new opportunities
for bio manufacturers to enter major markets around the globe. Biosimilar
versions of monoclonal antibodies have the probability to offer cost reductions
of 25-30%, and many emerging market countries are vigorously developing
pathways for approvals and are swiftly catching up. As this industry is
expanding the key biopharmaceutical players are adopting robust culturing
technologies to meet the increasing demand; thereby driving the growth of the
market.
Moreover, there is growing interest in improving the stem cell culture, because this technology is being used extensively in research for studying the stem cell biology, as well as for therapeutic applications. Furthermore, funding related to this research field has augmented in recent years which has accelerated the growth of the market. In addition to this, key media manufacturers launched new products for stem cell research. For instance, in September 2021, Bio-Techne Corporation launched a novel medium for the maintenance and expansion of induced pluripotent stem cells having applications in both translational and research workflows.
The outbreak of COVID-19 pandemic has improved the demand
for well-established cell-based vaccine production technologies. Moreover, it
has given rise to a few scientific innovations, particularly in the production
and testing of vaccine technology. For instance, the Vero line originated from
the African green monkey kidney and has been extensively used for viral vaccine
manufacturing. It has also been used for the development of various SARS-CoV
variants. ProVeroTM1 Serum-free Medium is one such medium manufactured by Lonza
Bioscience which is protein-free, and of non-animal origin designed to support
the growth of Vero cells and MDCK.
Moreover, in many European countries, cell-based flu vaccines
have been approved. A probable advantage of cell culture technology is that it
authorizes faster start-up of the manufacturing of vaccines during the
pandemic. Today, the development of superior biological models, the
optimization of culture growth medium, and the reduced dependence on
animal-derived components endure to drive the rapidly developing vaccine
development.
On the other hand, ethical issues concerning the use of
animal-derived products hinders the industry growth. For instance, FBS is
collected from the blood of fetal calves is one of the major ethical issues of
serum containing media. It is projected that 600,000 liters of FBS is achieved
from up to 1.8 million bovine fetuses are produced globally every year,
presenting momentous scientific and ethical challenges. To overcome this issue,
numerous workshops were held in the past on the replacement of fetal bovine
serum and possible ways to reduce the use of FBS in media.
Comments
Post a Comment